The IMO’s operating theatres obtain the biosafety classification required for cardiac interventions
16 de April de 2014
IMO’s eight operating theatres have just obtained the ISO 6 classification concerning the validation and qualification of rooms with controlled environments in hospitals. With AENOR’s (Spanish Association for Standardisation and Certification) new regulation, UNE 171340, ophthalmic operating theatres have gone from being considered ISO 7 (type B major ambulatory, conventional or emergency surgery) to ISO 6 (type A high-tech operating theatres, at the same level as those in which cardiac interventions or transplants are performed).
IMO is the first ophthalmology centre in Spain to obtain such a demanding classification
IMO is the first ophthalmology centre in Spain to obtain this new classification after its operating theatres underwent their annual validation by AENOR’s Standard 171 Indoor Environmental Quality Technical Committee on 5 April, which verifies compliance with biosafety requirements in surgical areas and serves to certify IMO’s commitment to the safety and welfare of its patients.
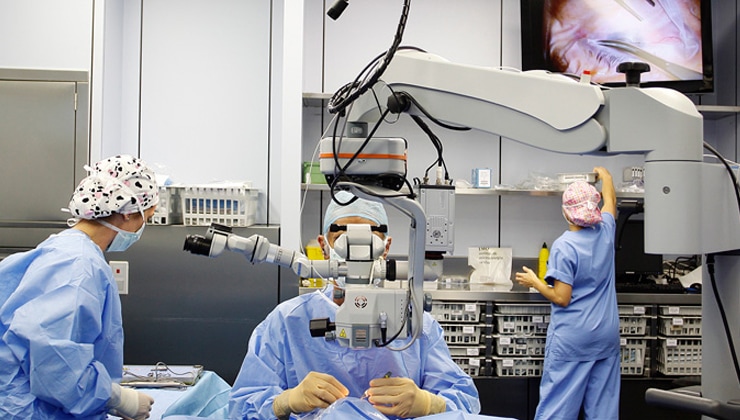
IMO’s surgical department is one of the most effective in Europe with regard to medical outcomes.
Maximum biosafety guarantees for any ophthalmic procedure
According to Marisa Jaén, head of IMO’s surgical department, “this new classification means more demanding and higher standards, which must be guaranteed even in seemingly simple and common surgical procedures, such as cataract operations, which also have to be performed according to strict requirements.” She adds, “IMO has actually already been fulfilling these requirements, since its aim with respect to its surgical department is not only to prevent the transmission of infections, of which there have been no instances since the new centre opened in 2009, but also to offer maximum comfort.”
Strict hygiene and prevention measures in the surgical department
According to the head of the surgical department, Marisa Jaén, some of the key aspects include “the spaciousness of the operating theatres, their highly technical equipment and strict work processes, maintenance and hygiene, a set of factors that make IMO’s surgical department one of the most effective in Europe in terms of medical outcomes, in addition to making it a leading centre internationally for comfort and safety.” The surgical department’s basic hygiene and prevention measures include the use of specific clothing (two-piece scrubs – tops and trousers –, caps, face masks, surgical shoes and leggings), surgical scrubbing to wash hands and forearms before entering the operating theatre and, after washing, inside the operating theatre, the wearing of sterile gowns and gloves for surgery. It is also important to note that the number of people who remain in the operating theatre during surgery should be restricted (only those that are strictly necessary) and that the instruments used should have undergone a prior process of cleaning, disinfection and sterilisation, while disposable materials used exclusively for each eye must be replaced by new ones after each procedure.
The spaciousness of the operating theatres, their highly technical equipment and strict work processes, maintenance and hygiene make IMO’s surgical department a leader internationally for comfort and safety.
“Attention to detail is so extreme that consideration is even given to providing plenty of room between elements in the work area to prevent the surgical team from brushing against furniture surfaces or even against each other, thereby ensuring maximum asepsis throughout the operation,” explains Marisa Jaén.
Facilities and technical systems for a controlled environment
In addition, as the operating theatre is a room with a controlled environment (one that requires optimum air quality, defined by concentration of particles), it needs to be equipped with facilities and technology specifically designed for it, such as a three-stage filtration system (large, medium and small particles). According to Jaén, “the installation of HEPA filters (high-efficiency particulate air), which prevent the passage of particles larger than 0.001 … 5 µm, such as bacteria and fungi, is one of the most important aspects to consider in the validation and qualification of rooms with controlled environments in healthcare centres.” Other key parameters in relation to biosafety, which determine the classification of these rooms, include: temperature (which must be kept between 20-24°C) and relative humidity (which has to be 45-55%), monitoring of environmental microbiology to detect and monitor the presence of microorganisms, and the control of noise, which should not exceed 40 dB to ensure comfort within the surgical department. Also essential, to ensure biosafety, are a pressure difference greater than 5 Pa between the “clean” or aseptic area (surgical department) and the “dirty” or septic area (outside the surgical department) and verification of the direction of the air to check that it is flowing from the “clean” to the “dirty” areas. Moreover, the ventilation system is designed to ensure a minimum of 20 air changes per hour and a flow rate of 1,200 m³/h, enabling the interior air to be renewed by the entry of exterior air, thereby contributing to improved environmental quality. Finally, it is important to ensure the “recovery of the room” through effective treatment of the air and allowing a certain amount of time to elapse between surgical procedures.
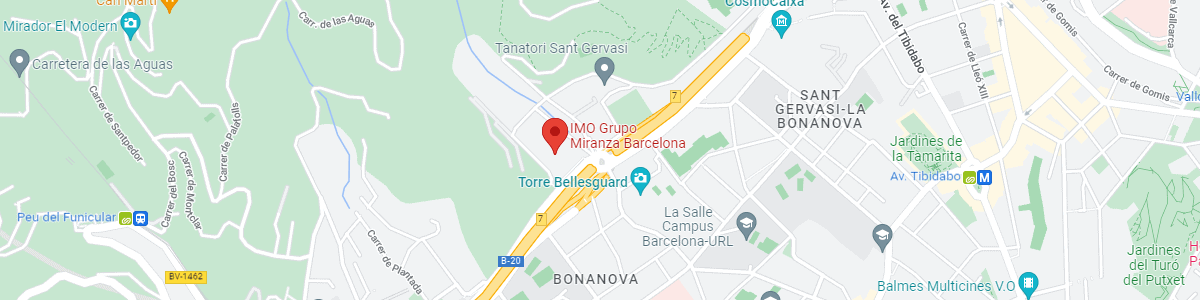
IMO Institute of Ocular Microsurgery
Josep María Lladó, 3
08035 Barcelona
Phone: (+34) 934 000 700
E-mail: international@imo.es
See map on Google Maps
By car
GPS navigator coordinates:
41º 24’ 38” N – 02º 07’ 29” E
Exit 7 of the Ronda de Dalt (mountain side). The clinic has a car park with more than 200 parking spaces.
By bus
Autobus H2: Rotonda de Bellesguard, parada 1540
Autobus 196: Josep Maria Lladó-Bellesguard, parada 3191
Autobuses H2, 123, 196: Ronda de Dalt – Bellesguard, parada 0071
How to arrive at IMO from:
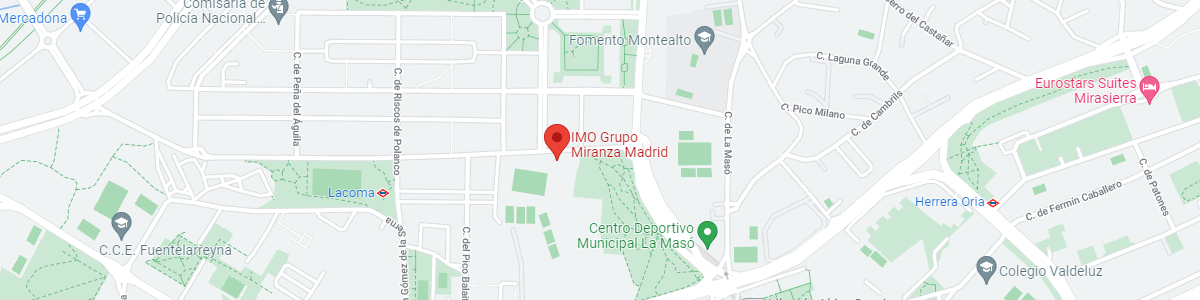
IMO Madrid
C/ Valle de Pinares Llanos, 3
28035 Madrid
Phone: (+34) 910 783 783
See map in Google Maps
Public transport
Metro Lacoma (líne 7)
Autobuses:
- Lines 49 & 64, stop “Senda del Infante”
- Line N21, stop “Metro Lacoma”
Timetables
Patient care:
Monday to Friday, 8 a.m. to 8 p.m.
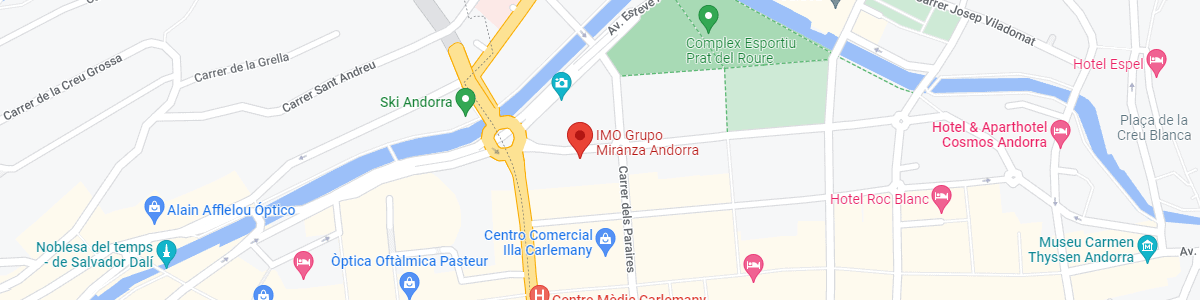
IMO Andorra
Av. de les Nacions Unides, 17
AD700 Escaldes-Engordany, Andorra
Phone: (+376) 688 55 44
See map in Google Maps
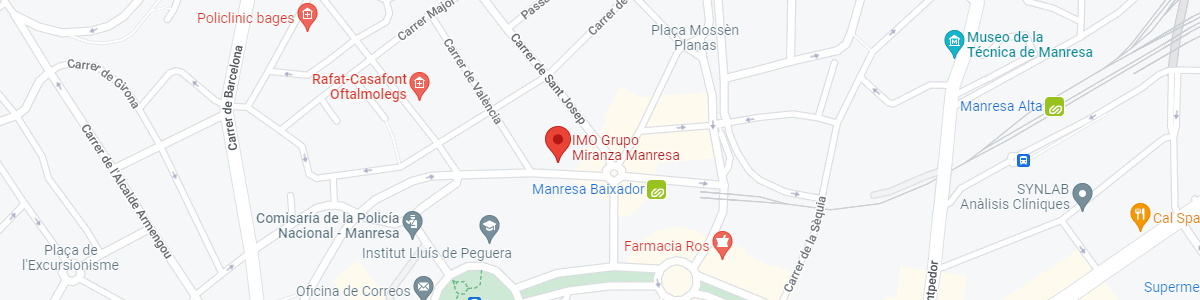
IMO Manresa
C/ Carrasco i Formiguera, 33 (Baixos)
08242 – Manresa
Tel: (+34) 938 749 160
See map in Google Maps
Public transport
FGC. Line R5 & R50 direction Manresa. Station/Stop: Baixador de Manresa
Timetables
Monday to Friday, 08:30 A.M – 13:30 PM / 15:00 PM – 20:00 PM