Genetic diagnosis and counselling at IMO Miranza Group
Dr. Esther Pomares explains what genetic diagnosis and counseling consists of.

What is genetic counseling?
Genetic analysis is performed on a blood sample, which is drawn from a patient affected by an inherited eye disease. The objective of this study is to identify the genetic alteration that triggers the pathology.
The objective of genetic analysis is to identify the alteration that causes the disease
What is an inherited disease?
Hereditary diseases usually have the following characteristics:
- are transmitted within the same family
- are caused by the alteration of a gene
- can follow different patterns of inheritance
The majority of inherited eye diseases are rare and affect approximately less than 1 in 2,000 people. At IMO Miranza group, we carry out the diagnosis and genetic counseling of more than 45 hereditary diseases that affect vision.
What is genetic diagnosis for?

Genetic diagnosis allows us to:
- Confirm the clinical diagnosis of the pathology, since some diseases present similar symptoms and / or can be difficult to diagnose.
- Know how the disease will evolve, since your prognosis may vary depending on the type of genetic alteration.
- Inform the affected family. To do this, a genetic counseling visit is carried out that aims to identify the most probable inheritance pattern, that is, the way in which the pathology could be transmitted in the family.
- Alert family members who may be carriers of the disease, as they could transmit it or suffer from it in the future.
- Prepare the affected patient for future gene and cell therapies.
- Better understand hereditary diseases and identify new responsible genes.

Should I have a genetic test?
We recommend that you request a genetic study if you meet one of these conditions:
- In your family medical history there are people with hereditary or recurrent eye diseases.
- You have been diagnosed with an inherited eye disease.
- You want to have children and, in your family or in that of your partner, there are cases of hereditary eye diseases.
- One of your children suffers from a hereditary eye disease
Pioneers in diagnostics and research
IMO Miranza Group Barcelona is the only ophthalmological center in Spain with its own molecular biology laboratory and a pioneer in the genetic diagnosis of hereditary eye diseases. In these facilities we carry out ambitious basic research projects, promoted by the IMO Foundation.
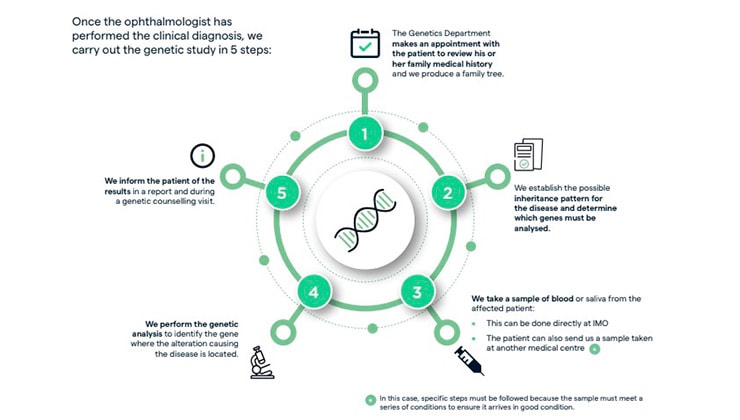
How is a genetic study performed?
Over 50 diseases, with more than 800 genes involved
These are the hereditary eye diseases for which MOI Miranza group provides a genetic diagnosis and counseling service:
- Achromatopsia · Alström Syndrome · AMD (age-related macular degeneration) · Aniridia · Anophthalmia · Anophthalmia ·
Autosomal dominant optic atrophy · Autosomal Recessive Bestrophinopathy · Axenfeld–Rieger syndrome - Bardet–Biedl syndrome Best disease · Blepharophimosis
- Central Areolar Choroidal Dystrophy (CACD) · Choroideremia · Cone dystrophy · Cone-rod dystrophy · Congenital cataract ·
Congenital stationary night blindness · Congenital fibrosis of the extraocular muscles · Congenital glaucoma · Congenital
hereditary endothelial dystrophy · Congentical and acquired ptosis · Congenital nystagmus - Donnai–Barrow syndrome
- Familial exudative vitreoretinopathy · Fuchs’ endothelial dystrophy
- Gyrate atrophy
- High myopia
- Joubert syndrome · Juvenile glaucoma
- Leber congenital amaurosis · Leber’s hereditary optic neuropathy
- Marshall syndrome · Meesmann corneal dystrophy · Microphthalmia
- Norrie disease
- Ocular albinism · Oculocutaneous albinism
- Primary open-angle glaucoma (familial)
- Reticular corneal dystrophy · Retinoblastoma Retinitis pigmentosa · Retinoschisis · Rothmund-Thomson syndrome
- Senior Loken syndrome · Sorsby dystrophy · Stargardt disease · Stickler syndrome
- Usher syndrome
- Wagner syndrome
More information
For more details, do not hesitate to contact us and/or download our genetic diagnosis and counseling guide.
IMO Institute of Ocular Microsurgery
Josep María Lladó, 3
08035 Barcelona
Phone: (+34) 934 000 700
E-mail: international@imo.es
See map on Google Maps
By car
GPS navigator coordinates:
41º 24’ 38” N – 02º 07’ 29” E
Exit 7 of the Ronda de Dalt (mountain side). The clinic has a car park with more than 200 parking spaces.
By bus
Autobus H2: Rotonda de Bellesguard, parada 1540
Autobus 196: Josep Maria Lladó-Bellesguard, parada 3191
Autobuses H2, 123, 196: Ronda de Dalt – Bellesguard, parada 0071
How to arrive at IMO from:
IMO Madrid
C/ Valle de Pinares Llanos, 3
28035 Madrid
Phone: (+34) 910 783 783
See map in Google Maps
Public transport
Metro Lacoma (líne 7)
Autobuses:
- Lines 49 & 64, stop “Senda del Infante”
- Line N21, stop “Metro Lacoma”
Timetables
Patient care:
Monday to Friday, 8 a.m. to 8 p.m.
IMO Andorra
Av. de les Nacions Unides, 17
AD700 Escaldes-Engordany, Andorra
Phone: (+376) 688 55 44
See map in Google Maps
IMO Manresa
C/ Carrasco i Formiguera, 33 (Baixos)
08242 – Manresa
Tel: (+34) 938 749 160
See map in Google Maps
Public transport
FGC. Line R5 & R50 direction Manresa. Station/Stop: Baixador de Manresa
Timetables
Monday to Friday, 08:30 A.M – 13:30 PM / 15:00 PM – 20:00 PM